Covid vaccine approved and will be rolled out across the UK from next week, Government confirms
and live on Freeview channel 276
The Department of Health and Social Care have revealed that Covid-19 vaccinations will begin across the UK from next week.
The Medicines and Healthcare products Regulatory Agency (MHRA) have approved the Pfizer and BioNTech jab for use in the country as part of a major breakthrough in the battle against the virus.
Advertisement
Hide AdAdvertisement
Hide AdAccording to studies, the vaccine has been shown to be 95% effective and works for all age groups.
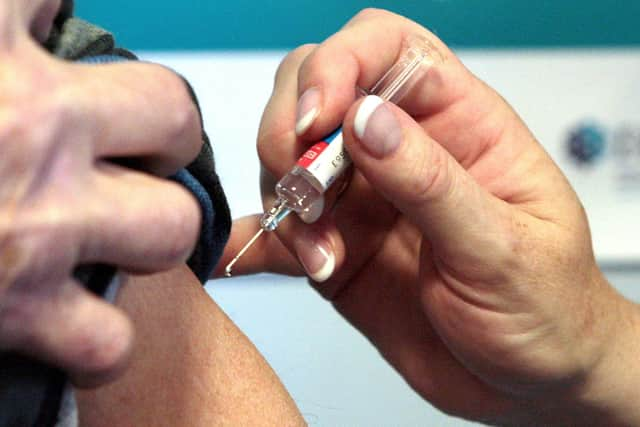

The Government has said that guidance will be published shortly on who will receive the vaccine first, however it is understood to be the elderly, care home residents, those working in the care sector and the clinically extremely vulnerable.
The Health Secretary, Matt Hancock, said that the UK was the first country in the world to have a “clinically authorised vaccine” to roll out.
Mr Hancock commented: “This is fantastic news. The MHRA, the fiercely independent regulator, has clinically authorised the vaccine for rollout.
“The NHS stands ready to make that happen.
Advertisement
Hide AdAdvertisement
Hide Ad“So, from early next week we will start the programme of vaccinating people against Covid-19 here in this country.”
A Department of Health and Social Care spokesman added: “The Government has today accepted the recommendation from the independent Medicines and Healthcare products Regulatory Agency (MHRA) to approve Pfizer/BioNTech’s Covid-19 vaccine for use.
“This follows months of rigorous clinical trials and a thorough analysis of the data by experts at the MHRA who have concluded that the vaccine has met its strict standards of safety, quality and effectiveness.
Advertisement
Hide AdAdvertisement
Hide Ad“The Joint Committee on Vaccination and Immunisation (JCVI) will shortly also publish its latest advice for the priority groups to receive the vaccine, including care home residents, health and care staff, the elderly and the clinically extremely vulnerable.
“The vaccine will be made available across the UK from next week.”